Atoms and
Molecules...
do they matter?
Introduction:
They are everywhere - in the floor beneath your feet, in the air around you, and yes, even inside you. We are all just a big bunch of atoms and molecules. What do these particles have to do with the states of matter, and how do they explain the changes from solid to liquid to gas? You are going on a quest, like the knights of old. But in this case you will be searching, in books and on the internet, to find answers to these questions. You may even see some interesting sights along the way.
The Task:
Working independently, you will:The Process:
Matter
"
The first question we need to ask is probably, "What is matter?" Click on matter and your first stop on this quest will give you a pretty good idea of what we are talking about. Then, click on the window button, then page one and join the lesson again.
Answer the following questions in your notes (look back at the site if you need to):
1. What is matter?
2. What are the 5 states of matter?
3. What is a compound?
Now you have completed the section on matter, click on matter pop quiz and see how well you can answer the questions. You may work with your partner. After each question, it will tell you if you were correct or not. Record you score on a sheet of paper.
An activity that will give you some idea of just how very small atoms and molecules are, can be found by clicking on tiny.
You can print out the instructions, and do this activity with your partner at your desk. Measure your paper after each cut, and compare your measurements with the ones you will find in the activity. After you have finished playing with this activity, come back to the computer.
States of Matter
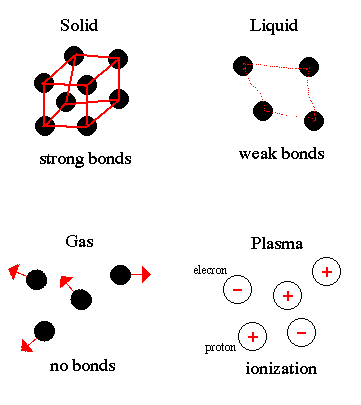
Now you have some idea of how very small atoms and molecules are. No wonder adding just a little heat can cause them to speed up and move farther apart, to give us solids, liquids, gasses, and plasma. For another explanation of states of matter, click on states.
Now you have completed the section on states of matter, click on states pop quiz and see how well you can answer the questions. You may work with your partner. After each question, it will tell you if you were correct or not. Record you score on a sheet of paper.
The first state we will be investigating is the solid state. Most of the things around us are solids. Anything that has a constant shape and size, like a house, a chair, or even this computer, is mainly solid. Some of the facts we have learned about molecules can help to explain this. For more details and some interesting pictures about the structure of solids, click on solids.
Answer the following questions in your notes (look back at the site if you need to):
1. Why are solids hard?
2. What is one physical characteristic of a solid?
3. What is a mixture?
The next state we will visit is the liquid state. We see lots of liquids every day: the drinks we drink the water we swim in, bathe in, and the rain that falls on us. These materials have a definite size- one cup of water is always one cup of water, even if you pour it into something else- but they have no definite shape. The water coming out of the faucet soon takes the shape of the glass you pour it in. Click on liquids.
Answer the following questions in your notes (look back at the site if you need to):
1. Why are gases an in-between state of matter?
2. What are the physical characteristics of a liquid?
3. What is the name of the forces at work that keep liquid molecules together?
Gasses make up, a third state of matter, but one that is not so easy to discuss, because most gasses are invisible. The air we breathe is a good example. We can't see it, or feel it, unless the wind is blowing or we are moving fast, but we know it's there. A gas doesn't have a definite shape- air from our lungs can fill up a balloon. And it doesn't have a definite size. Liquid that evaporates from a drop of perfume can fill up an entire room. The movement of molecules has something to with this also. Click on gasses.
Answer the following questions in your notes (look back at the site if you need to):
1. Why are gasses so spread out?
2. What are the physical characteristics of gasses?
3. Give an example of how you use compresses gasses every day.
The last state of matter is plasma. This is really different from the others, and not one you see much on earth. Click on plasma.
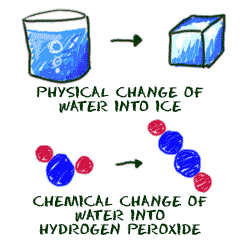
Now that you have visited the different states of matter, it's time to talk about how they change from one to the other. If you think about the changes of state that occur around us most often, the change of water from ice to water to steam, it will probably occur to you that one thing that plays an important role in these changes is temperature. This makes sense, when we think of the fact that molecules speed up when heat is applied, and that the movements of these molecules, and their distance apart, is what determines whether something is a solid, a liquid, or a gas. What may surprise you is that all sorts of materials undergo these changes. Even metals like iron, if heated enough, can melt. For an interesting activity using these facts, click on temperature and follow the instructions.
Answer the following questions in your notes (look back at the site if you need to):
1. What happens to the elements or molecules as the temperature changes?
EvaluationNow that you have become an expert on the phases of matter, it is time to share your knowledge with the class. You will prepare a brochure, representing the molecules in the different phases of matter. Each state of matter (Solid, Liquid, and Gas) will be on a different diagram. Be sure to: